People use artificial tear drops every day to combat dry eye conditions. The cause of dry eyes varies from person to person, and using artificial tears is often recommended to lubricate the eyeball, providing much-needed relief. Causes of dry eyes can include specific medications, health conditions, and other environmental factors. Dry eye symptoms can consist of feeling something in the eye, burning pain, blurred vision, light sensitivity, and mucus production. These symptoms all impact a person’s quality of life. Treating dry eyes with over-the-counter artificial eye tears can help make people more comfortable.
In December, some who use artificial tears to combat dry eyes began to develop infections. The source is believed to be an extensively drug-resistant strain of Pseudomonas aeruginosa, which has never been reported in the U.S. before now.
An investigation conducted by the CDC and the FDA identified artificial tears as a potential source of exposure. More than 67 patients in 16 different states have been affected by infections caused by this bacteria. Three people passed away, eight lost vision in at least one eye, and four had surgically removed eyeballs due to the condition. This bacterial strain is drug-resistant, meaning the antibiotics typically used to treat similar bacterial eye infections are ineffective.
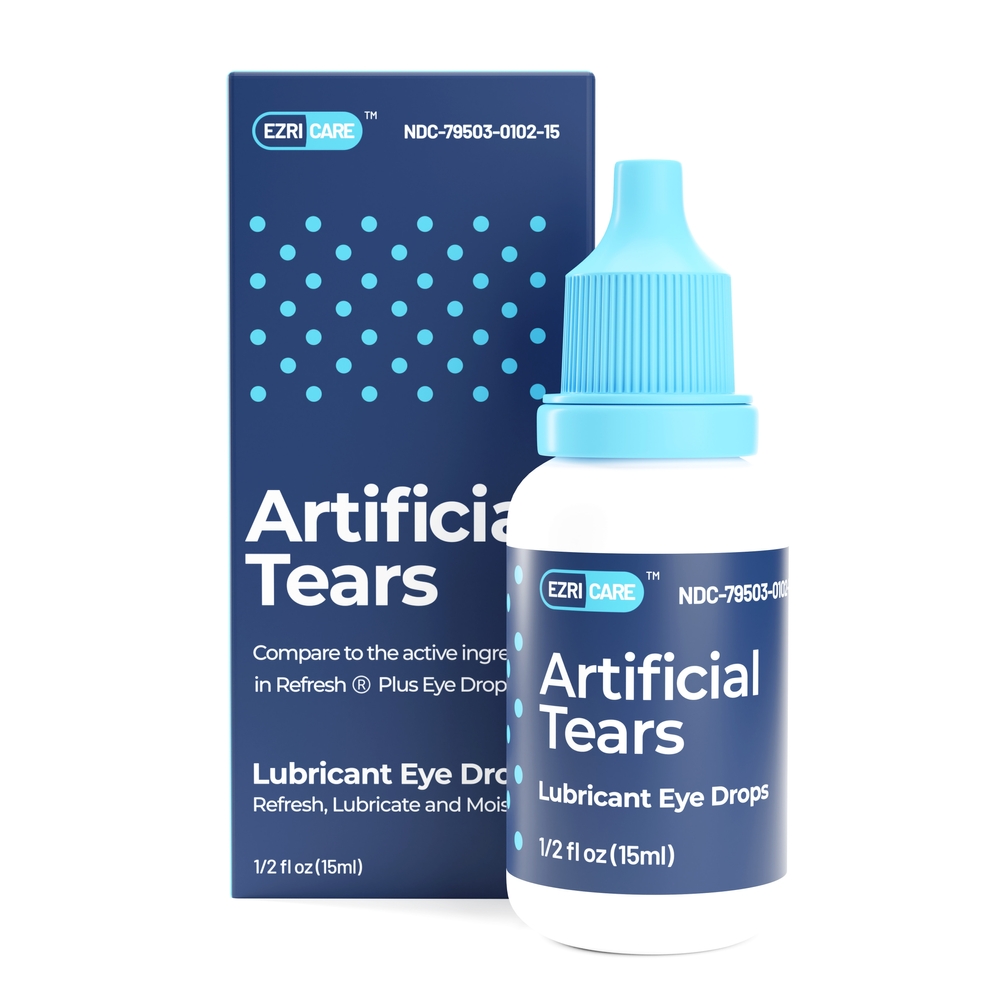
The FDA issued a warning to stop using specific eye drops as there were suspicions that eye drops could contain dangerous bacteria. On February 2, Global Pharma issued a voluntary recall that eye drops sold as EzriCare Artificial Tears Lubricant Eye Drops and Delsam Pharma Artificial Tears Lubricant Eye Drops were recalled due to potential contamination with bacteria. On February 21, the voluntary warning and recall expanded to include Delsam Artificial Eye ointment. These types of eye drops are commonly sold online through retailers like Amazon.
It is believed that the artificial tears eye drop recalls came about because the manufacturer failed to adequately test the product for bacterial contamination without adequate preservation. This violation of the Current Good Manufacturing Practice requires that products used directly on the conjunctival sac, conjunctiva, or on eyelids be sterile and manufactured in a sterile environment.
This news can be worrisome if you or a loved one has used EzriCare or Delsam Pharma’s artificial tears. If you have experienced any infection symptoms, stop using these brands of artificial tears and seek medical attention. Infections caused by the Pseudomonas aeruginosa bacteria are aggressive and difficult to treat. Medical treatment is required to treat this type of infection. After seeking medical treatment, consider exploring legal avenues by consulting an attorney.
Symptoms of Artificial Tears Infection
While infections are concerning, those associated with the eye are more worrisome. As many depend on their eyesight for navigating the world, contracting a potentially devastating infection is anxious news. Symptoms of eye infections can include the following:
- Eye discomfort or pain
- Clear, green, or yellow discharge from the eye
- Blurry vision
- Increased sensitivity to light
- Feeling of something in the eye (a sensation of a foreign body in the eye)
- Redness of the eyelid or the eyeball
Under normal circumstances, the Pseudomonas bacteria can be found commonly in water and soil. When it causes infection in people, Pseudomonas aeruginosa can cause infection in the lungs, such as pneumonia, and in the blood and other body parts after surgery. This bacteria “can destroy the eye within 48 hours,” according to Darlene Miller, head of Bascom Palmer Eye Institute laboratory.
While some patients with dry eye conditions have stopped using EzriCare or Delsam Pharma’s artificial tears, others may not have. Doctors, including Dr. Guillermo Amescua, a cornea specialist at Bascom Palmer Eye Institute of the University of Miami Miller School of Medicine, believe that eye infections will continue to develop until everyone stops using these contaminated eye drops.
The first EzriCare eye drops lawsuit was filed in Florida in early February. Filed in the U.S. District Court for the Middle District of Florida, the plaintiff alleges that she developed an eye infection after using the EzriCare artificial drops she purchased at Walmart. The plaintiff required surgery when antibiotics did not clear the infection. Walmart is also named as a defendant in the lawsuit, which is believed to be the first of its kind.
If you or a loved one has used EzriCare and Delsam eye drops and ointment and developed symptoms of an eye infection, contact your medical professional for treatment. Do not hesitate to do so. In the case of this bacteria, time may mean vision. Delaying treatment may result in vision loss. Also, seeking advice from an attorney with expertise in personal injury cases will provide legal options to explore. Because of the diverse nature of infections, you may be eligible for compensation for lost wages, medical bills, and pain and suffering.
Using eye drops to treat dry eyes should not cost people their vision or the use of their eyesight. In the case of EzriCare and Delsam eye drops and ointment, using these products may have cost consumers the gift of seeing the world.